Chromatography Making water pure (Distillation) Purifying rock salt
Something is a MIXTURE if it contains two (or more) substances that are not chemically joined together.
The substances in a mixture can often be easily separated from one another.
A PURE substance only contains one material and so cannot be separated in any way (unless a chemical reaction takes place)
Summary of methods used to separate a mixture
| PROCESS | What it is used for | Examples |
| Using a magnet | Separating something containing iron from another material | Separating iron filings from sandThe iron is attracted to the magnet |
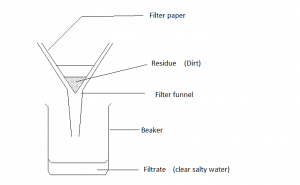
| Filtration | Used for separating a solid or suspension from a liquid | eg separating SAND from waterThe particles of sand are too large to fit through the holes in the filter paper. |
| Evaporation | Used to obtain the solute from a solution | eg for obtaining SALT from salty water The water evaporates leaving the salt behind |
| Simple distillation | Used to obtain the solvent from a solution | eg for obtaining pure water from sea water.The water boils and urns into steam. The steam is allowed to condense back into pure water which flows into another container |
| Fractional distillation | Used to separate one liquid from a mixture of different liquids that have different boiling points. | eg i. obtaining ALCOHOL (bp78C) from wine. ii. Oxygen or nitrogen is obtained from air (a mixture of different gasses) by the fractional distillation of liquid air.Similar to simple distillation but because the alcohol boils at a lower temperature than water is boils away before the water. |
| Chromatography | Used to separate out one colour from a mixture of colours | eg for separating out the colours in black inkColoured inks are carried up a piece of filter paper by a suitable solvent,Some colours travel quicker than others which causes them to separate. |
Two immiscible liquids (liquids that don’t mix like oil and water) can be separated using a separating funnel.
The more dense liquid sinks to the bottom and can be removed by opening the tap.
 MAKING ROCK SALT PURE
MAKING ROCK SALT PURE
Rock salt is a MIXTURE of several substances.
In order to make pure salt it is necessary to remove the INSOLUBLE dirt from the SOLUBLE salt.
[Note: The method described below could be used to separate any two substances when one of them can dissolve in water and the other cannot]
1. CRUSH the rock salt using a mortar and pestle. This makes the salt dissolve more easily.
2. STIR the crushed salt into a beaker of warm water. The salt will DISSOLVE into the water but the dirt, which is insoluble in water, will not.
3. FILTER the mixture. The dirt will get caught in the filter paper as a residue and the clear SALTY WATER will drip through and form the filtrate.
4. Warm the filtrate in an evaporating basin. The water will EVAPORATE leaving behind pure salt crystals. The heat is stopped BEFORE all of the water has evaporated. This makes the salt more pure and helps prevent the evaporating basin from cracking..
The last little bit of salty water is filtered out
Water can be purified by a process called DISTILLATION.
eg: If we wanted to make some PURE WATER from SEA WATER we would distill the sea water.
The beaker of cold water helps keep the test tube cold so more steam condenses
This method to make pure water shown above is ok but a more efficient method is shown below:
The water in the flask EVAPORATES and turns into steam.
Anything dissolved in the water cannot evaporate and so remains in the flask.
The steam CONDENSES in the delivery tube and turns back into water which collects in the test tube.
How to tell if a liquid is pure or contains a solute:
- Place one or two drops of the filtrate on to a clean watch glass (or microscope slide)
- Let the water evaporate and see if there is any residue.
- If there is it means that some of the solid did dissolve.
(back to Top)
Chromatography
Chromatopgraphy is a way of separating a mixture of coloured dyes or pigments such as found in ink.
For example the green ink found in a green felt tip pen might be made of a mixture of blue and yellow ink and chomatography will show us which colours were actually used.
How to make a chromatogram
Place a drop of the coloured ink being tested near the end of a strip of chromatography paper (or filter paper) and suspend the paper in a suitable solvent (usually water but other solvents such as alcohol can be used).
The spot of ink is placed on near the end of a piece of chromatography paper (a bit like filter paper) wich is lowered into a glass jar or tank (which i covered to stop the solvent evaporating).
The solvent rises up the paper carrying the coloured dyes with it. Some dyes travel quicker than others which cause them to separate (fgure 2)
Often severall inks are placed on the same filter paper which allows us to compare the dyes used in different inks. (Figure 3)
Coloured spots at the same level (like the green dots) mean that the same green dye was used in both inks. The ink on the left contaiuned no green dye.
Words to know:
Mixture: Something that can be SEPARATED into two (or more) different substances. eg air, sea water, earth, orange juice.
Pure: A material is pure if it contains only one chemical substance and so cannot be separated further
Filtrate: The clear liquid that has been filtered.
Residue: The solid left behind after an experiment (eg the solid left in the filter paper).
Filtration: The process used to separate a solid (or suspension) from a liquid.
Suspension: Very fine particles of solid mixed with a liquid. If the solid is in suspension the water will often look cloudy. eg flour and water shaken together.
NOTE: in a suspension the solid has not dissolved but in a solution it has.
Soluble: Can dissolve
Insoluble: Cannot dissolve
Solvent: The liquid that is used to make a solution
Solute: A substance that has been dissolved in a solution.